Published: Mar 11, 2019Time to read: 9mins Category: Learning
4 Steps to Take to Prepare for a Compliance Audit
Looking to prepare for a compliance audit? In this blog from Nancy Dietz, Customer Success Manager, we explore a few ways your LMS can help you impress auditors and inspectors.
Healthcare compliance inspections can return any number of findings. Some are simple suggestions to fix small points that can make day-to-day life at work easier. And others can result in severe consequences if they’re not immediately acted on in the right way.
There’s no avoiding the fact that a compliance audit is a big deal. Too many very severe findings can see hospital systems suffer penalties and lose accreditation from governing bodies, often with harsh financial and reputational implications.
With almost all organizations seeking to improve their audit trail for regulators, here are four ways to face the compliance audit challenge with confidence.
How to Prepare for a Compliance Audit Step #1: Be Prepared for Surprises
Depending on the regulatory body, there is often a window of time in which auditors can arrive for an inspection without a specific scheduled appointment. Even when companies are forewarned that an inspection will take place within a set period (known as a survey window), it could happen on the first or last day of that window. Regulators can also decide to carry out an inspection in response to an incident, such as a death or a problem reported by a hospital visitor.

Depending on the size of a facility and what needs to be surveyed, an inspection can last less than a day or several days. Surveyors use what is called the “tracer methodology”. This process will trace a patient’s visit throughout the system. This allows for a surveyor to be assured all procedures are being followed and that caregivers are properly qualified. Practice tracers are often held by organizations to identify any gaps in procedures.
Regulators such as The Joint Commission (TJC), who identify concerns such as putting the wrong labels on samples in a laboratory, can also set these against standards such as the CLIA, setting off a separate review.
Waiting for a regulator to announce a survey window—or scrambling to piece everything together in the build-up to an inspection—is a recipe for non-compliance. Successful organizations consistently achieve full compliance by anticipating the full scope of questions regulators could ask at any time—and making sure they use powerful technology, like a learning management system, to help them remain ready for anything.
So how can an LMS support this always prepared approach? Your LMS should be able to:
- Track the distribution of procedure(s)
- Track an employee acknowledgment and understanding of procedure(s)
- Track qualifications related to either internal or external certifications/education
- Remind employees when it’s time to renew training
- Auto-enroll employees when they’re due for refresher training
- Provide visibility for managers or other reviewers to assure records are complete
Recommended related reading: 'The 13 Must-Have Features of a Learning Management'
How to Prepare for a Compliance Audit Step #2: Keep Everything in One Place
At the start of an on-site visit, surveyors meet with all of the relevant executives and management staff to identify the agenda that will be followed during the visit.

They’re then typically given a stack of patient records, from which they pick out and read through a few. Keeping these records correctly requires a comprehensive, centralized approach. Patient records are stored in an electronic medical record system (EMR), also sometimes known as an electronic health record (EHR). The same centralized records keeping is necessary for employee education and qualifications.
Auditors and surveyors are always unimpressed when compliance staff cannot locate and produce accurate reports for employee training and qualification. For example, when a surveyor traces the journey of a patient through the emergency department, they may want to know how the employees are trained to protect patient identifier data, or how the employee is qualified to take initial health statistics. If the patient access checked them in and verified insurance, are they trained to do this? Do they have any patient access certifications? These types of questions are reviewed for every employee that interacted with the patient during their stay.
An organization with an inefficient compliance management system might find that the data in each instance was kept in multiple locations. For example, the patient identifier training was through an eLearning course, while the certification information is on paper in the unit manager’s office file. Sometimes, the data is known but never documented officially or perhaps even lost.
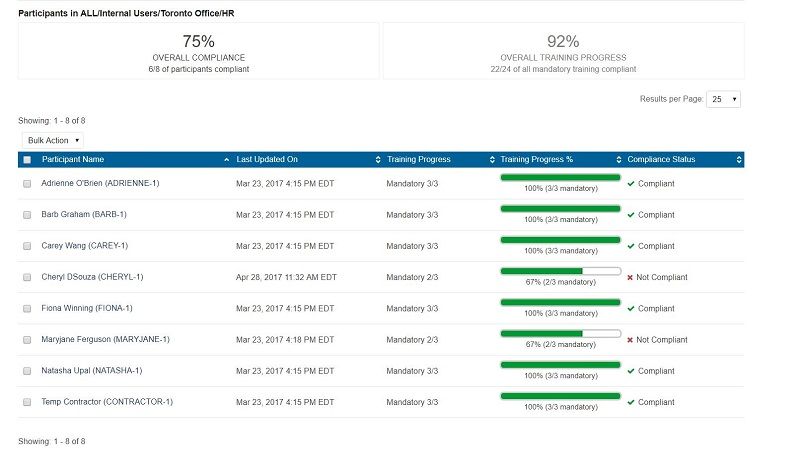
Removing this risk requires the use of an LMS that records and stores all of the data needed for inspection in a central resource that is accessible to all compliance staff and ready to produce rigorous reports at any time.
How can the LMS help support this centralized storage of training and qualifications?
- Internal courses are recorded to a transcript with assessment scores and completion dates
- Certifications acquired either within the organization or externally can be tracked with award dates and expiring dates
- Reminder messaging can be sent to remind employees to refresh training or even to renew professional memberships
- Scanned documents can be uploaded and stored within the LMS to avoid the loss of originals
- Reports can be generated by anyone with permission, avoiding locked offices or confusion about where documentation is being stored
- Reports generated are real-time data, not waiting for an overnight update or even an hourly refresh of data
More from the blog: 'How a Compliance Training Solution Helps You Avoid Common Audit Errors'
How to Prepare for a Compliance Audit Step #3: Align Internal Rules With Regulatory Standards
Organizations need to be able to demonstrate that staff know how to follow rules set by groups such as The Joint Commission, by generating reports capable of illustrating that the people concerned are aware of the rules and know the steps to follow those rules.

But they also need to be able to demonstrate how they’re educating staff on internal policies and procedures, and show that the relevant people have the knowledge to implement them. An example of this is the creation of weapons policies by organizations. Staff need to address a weapon in the Emergency Department carried by an on-duty officer differently than an off-duty officer or patient. There is also a different response for an active shooter scenario.
Each requires an understanding of:
- the organization policy
- the steps that a staff member is expected to perform for their protection
- and their patient’s protection
As with regulatory requirements, setting a training routine is the best way to ensure staff are fluent in the latest company policies and procedures. When learning takes place at set intervals, such as annually, any changes in policies can be recognized and reflected in employee training. Just prior to the rollout of annual compliance, a thorough review of content should be performed with the assistance of subject matter experts (SMEs). If a regulation changes after rollout, then all employees must be trained on the most recent and accurate content.
How does the LMS support this alignment?
- Dates can be added to courses to identify which ones are due for review within a month, quarter or year
- Revision control to identify which release an employee received their training on
- Interim content updates can be uploaded and available immediately for all new enrollments, plus anyone who’s not already completed the training
- Interim content updates can also be re-assigned to employees that completed the earlier release with a notification advising of the need to complete the course again
You might also like: 'Managing the Learner Journey With LMS Login Notifications'
How to Prepare for a Compliance Audit Step #4: Everyone Has Compliance Responsibility
Imagine a surveyor walks through a hospital hallway during an inspection and randomly asks a student nurse what they would do if they heard the fire alarm. The nurse has only been at the hospital for a few days as part of their studies, but the initial compliance training prepares the student to clearly demonstrate the appropriate health and safety knowledge. This student’s understanding of the procedure in the event of a building fire becomes one of the findings of the inspection report.
Whether they’re full-time or casual workers, executive or support team members, surveyors hold all staff equally responsible for adhering to standards. Employees also need to be fully compliant across all of the environments they might visit within an organization and all of the specific requirements of their role.
Some examples:
- If a piece of electrical equipment in a laboratory fails, maintenance staff who might spend most of their professional lives in a very different setting need to know how to enter the facility and complete the repair while remaining compliant.
- A clinician who uses a surgical suite infrequently is expected to be as versed in compliance as a permanent employee while they’re on-site.
From records of training carried out externally to alerts when contractors are about to require re-training, the right LMS helps organizations to manage this network at scale.
Your LMS is Critical to Compliance Success
As we’ve seen, there are many steps to take when preparing for a compliance audit.
Tracking compliance can be a complex business. For L&D teams at healthcare organizations with large workforces consisting of both employees (eg. full-time, part time) and non-employees (eg. students, contractors, credentialed practitioners), ensuring that everyone has taken the right training—and proving it—can be a major challenge.
The secret to success is finding a robust, adaptable LMS that updates records in real-time. With this essential element, you can ensure complete compliance and be inspection-ready all year round.
Discover How Learning Builds Skills and Ensures Compliance
Design, deploy, track, analyze, and report on enterprise learning and compliance programs. PeopleFluent helps you execute your programs seamlessly, so employees upgrade their skills and you get results.